Scope of mono, di-, tri-, and tetrasubstituted aromatic olefins

Scope of mono, di-, tri-, and tetrasubstituted aromatic olefins

Dual Rh−Ru Catalysts for Reductive Hydroformylation of Olefins to Alcohols - Rodrigues - 2018 - ChemSusChem - Wiley Online Library

Recent Advances in Mono‐ and Difunctionalization of Unactivated Olefins - Patel - 2021 - Asian Journal of Organic Chemistry - Wiley Online Library

Stereoselective Synthesis of Tri- and Tetrasubstituted Olefins via 1,6-Additions of Diazo Compounds and Their Precursors to p-Quinone Methides
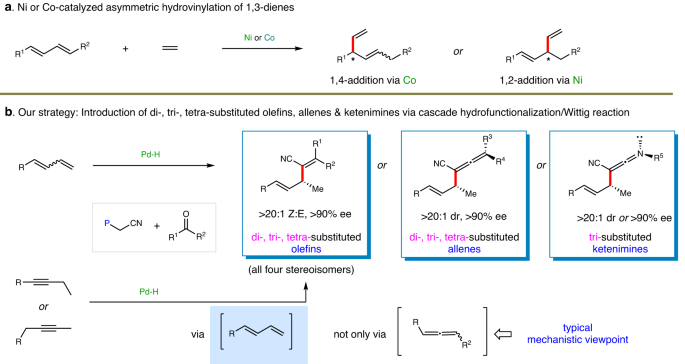
Asymmetric formal sp2-hydrocarbonations of dienes and alkynes via palladium hydride catalysis
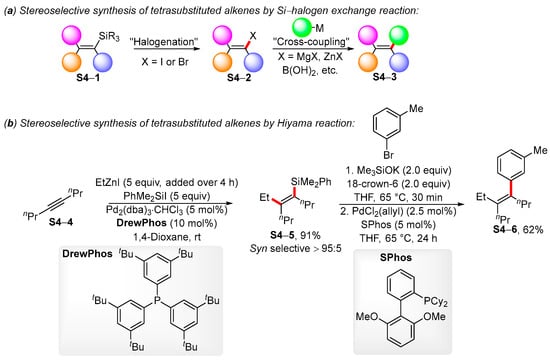
Organics, Free Full-Text

Strategies in Pd-Catalyzed Dehydroaromatization (DHA) a Conventional

Stereoselective Synthesis of Tri- and Tetrasubstituted Olefins via 1,6-Additions of Diazo Compounds and Their Precursors to p-Quinone Methides

Base-free NiH-catalyzed regio- and stereo-selective hydroacylation of allenes: A new route to synthesis of tetra-substituted olefins - ScienceDirect

Enantioselective nickel-catalyzed dicarbofunctionalization of 3,3,3-trifluoropropene
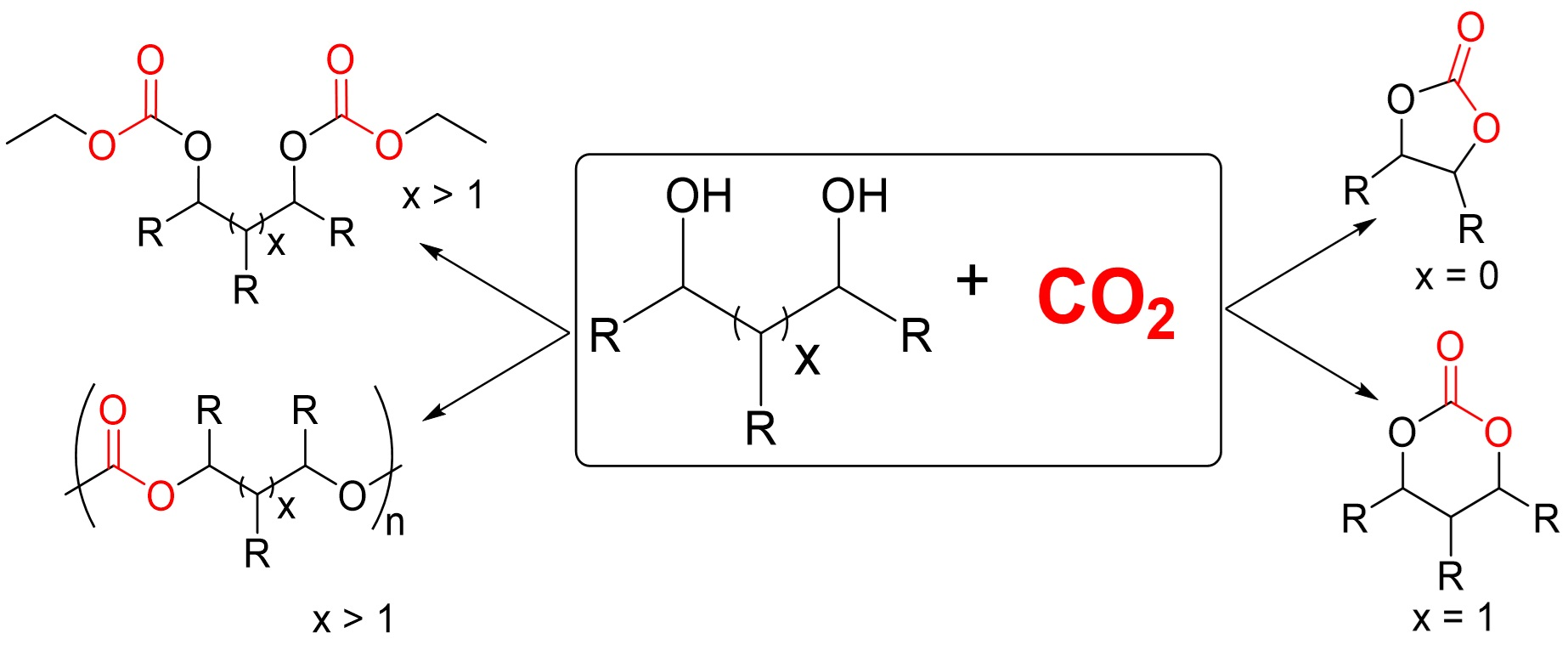
Catalysts, Free Full-Text

Scope of mono, di-, tri-, and tetrasubstituted aromatic olefins

Stereoselective Synthesis of Tri- and Tetrasubstituted Olefins via 1,6-Additions of Diazo Compounds and Their Precursors to p-Quinone Methides

Scope of mono, di-, tri-, and tetrasubstituted aromatic olefins