a)Adsorption energy comparison between twice of * NO and N-N

Download scientific diagram | (a)Adsorption energy comparison between twice of * NO and N-N coupling intermediate * ONNO. The horizontal line shows the equilibrium between 2NO(g) and 2 * NO, while the diagonal line illustrates the equal adsorption energy between 2 * NO and * ONNO. (b)The activation barriers for N-N coupling between * N and NO (gas) vs * N and * N. where the unfilled markers show around 0.44 Monolayer of NO coverage is considered. (c) Adsorption energy comparison between * NO and * N 2 O.The horizontal line shows the equilibrium between NO(g) and * NO while the vertical line depicts the equilibrium between N 2 O and * N 2 O under standard conditions. from publication: Electrochemical Nitric Oxide Reduction on Metal Surfaces | Electrocatalytic denitrification is a promising technology for removing NOx species (NO3− , NO2− and NO). For NOx electroreduction (NOxRR), there is a desire for understanding the catalytic parameters that control the product distribution. Here, we elucidate selectivity and | Nitric Oxide, Electrochemistry and CO2 Reduction | ResearchGate, the professional network for scientists.

Two-dimension Cd-N co-doped α-AsP as high sensitivity and selectivity NO and NO2 sensor: A first-principles study - ScienceDirect
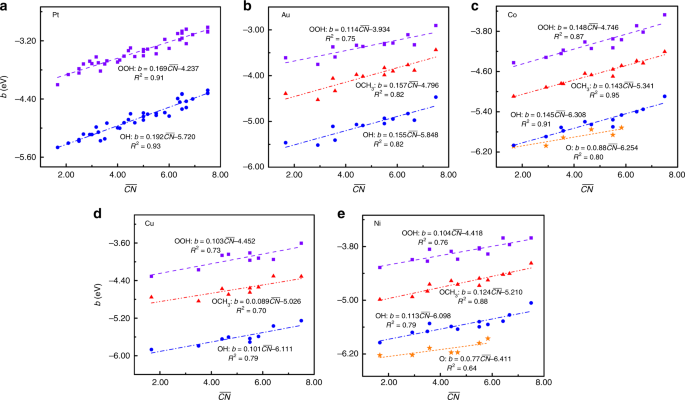
Determining the adsorption energies of small molecules with the intrinsic properties of adsorbates and substrates

Comparison between adsorption enthalpy and Gibbs free adsorption energy

Adsorption Energy - an overview

a)Adsorption energy comparison between twice of * NO and N-N coupling

A Linear Relationship between the Charge Transfer Amount and Level Alignment in Molecule/Two-Dimensional Adsorption Systems

Adsorption energy E ads defined as the difference between the
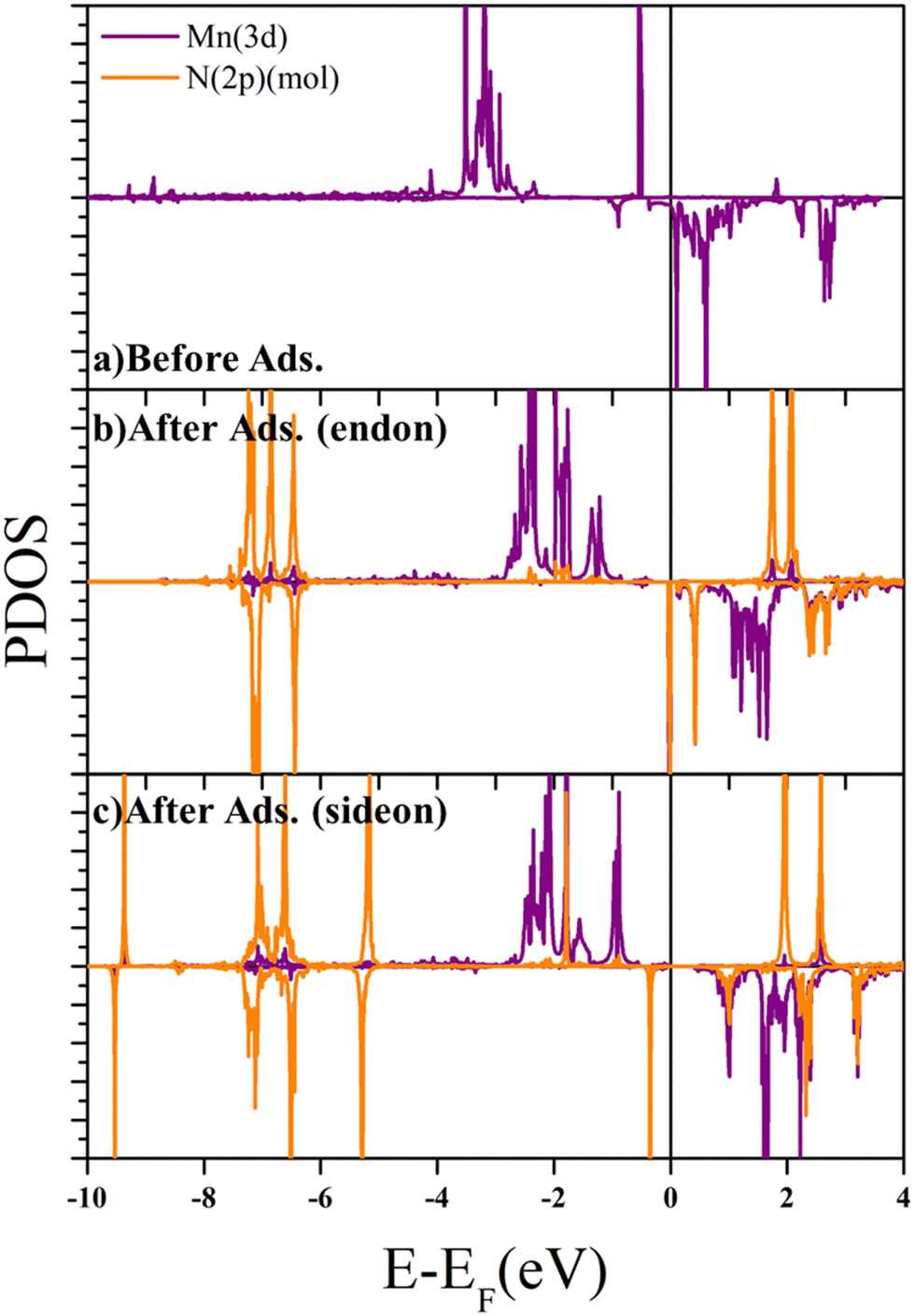
Adsorption mechanism of the N 2 and NRR intermediates on oxygen modified MnN 4 –graphene layers – a single atom catalysis perspective - Physical Chemistry Chemical Physics (RSC Publishing) DOI:10.1039/D2CP05491D

Adsorption energy E ads defined as the difference between the

Adsorption energies, distances and bader charge analysis of (100) surface.

Adsorption configurations of NO, NO2 and N2O onto intrinsic graphene
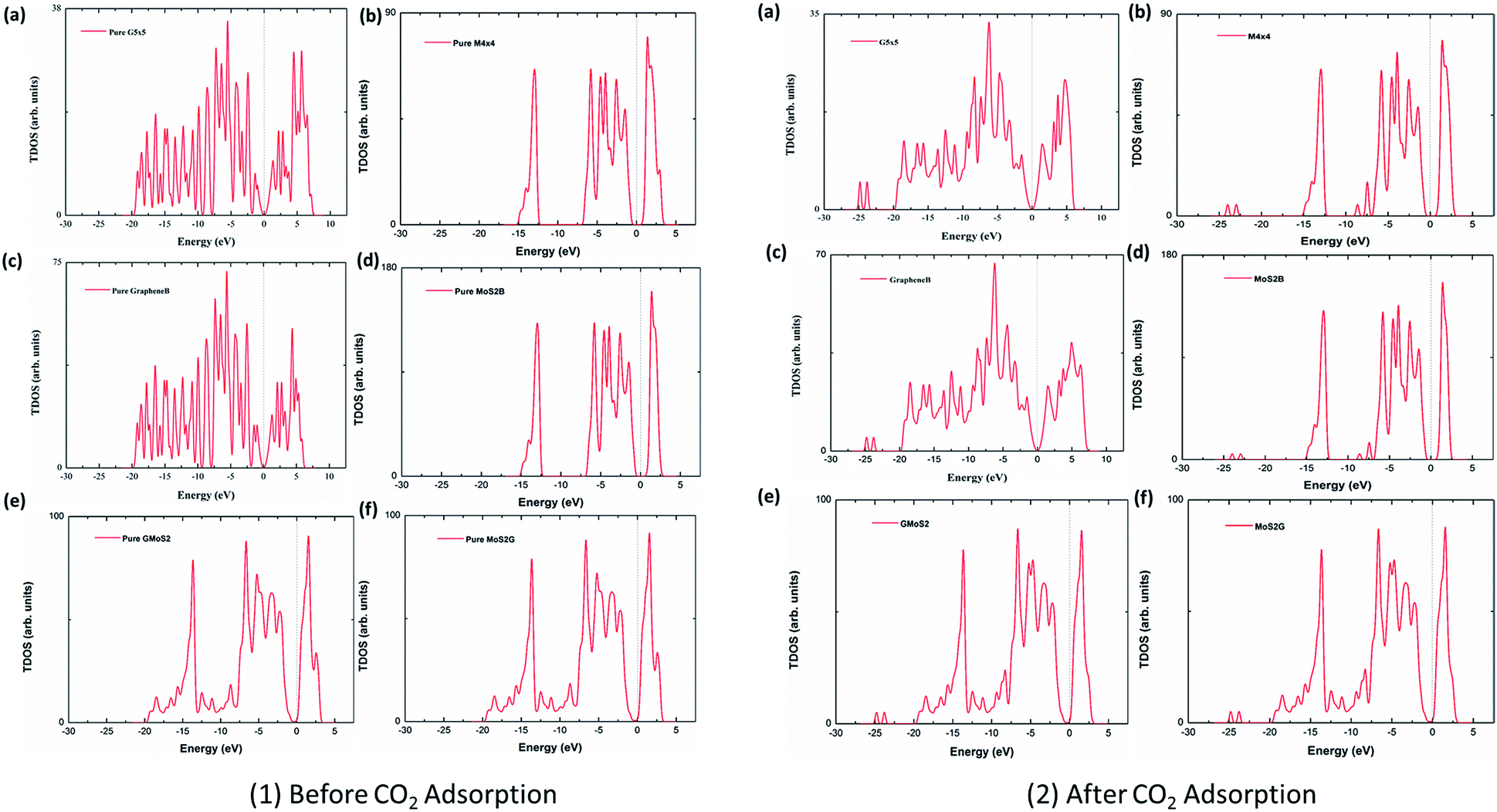
A comparative study of mechanisms of the adsorption of CO 2 confined within graphene–MoS 2 nanosheets: a DFT trend study - Nanoscale Advances (RSC Publishing) DOI:10.1039/C8NA00314A