Is CH3OH (Methanol) Soluble or Insoluble in Water?


SOLVED: Which of the following liquids is least soluble in water

Is CH3OH (Methanol) an Electrolyte or Non-Electrolyte?

Which is more polar: H2O or CH3OH? - Quora

Explain why Methanol (CH3OH) is soluble in water but Benzene (C6H6

Solved B. Solutes and Solvents 1. Solubility SOLVENT water
Why is methanol miscible in water while iodomethane is not? - Quora
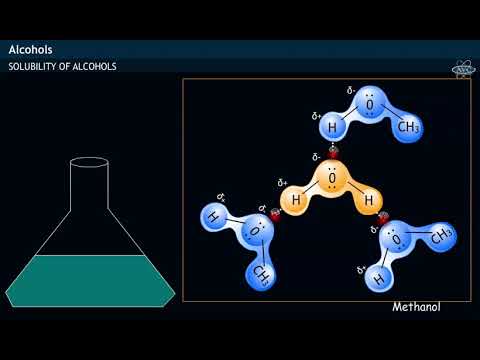
How does methanol soluble in water ? SOLUBILITY OF ALCOHOL

Solubility and Density Isotherms for the Sodium Sulfate−Water

Solved Which of the following statement(s) about the
Slides30a

Three Solvents: Water, Methanol, Cyclohexane PAP
Is CH3OH soluble in water but is CCL4 insoluble? - Quora

Question Video: Identifying the Alcohol That Is the Least Soluble