Methanol, Structure, Density & Molar Mass - Video & Lesson Transcript


Methanol vs. Ethanol: What's the Difference?
Methanol CAS 67-56-1
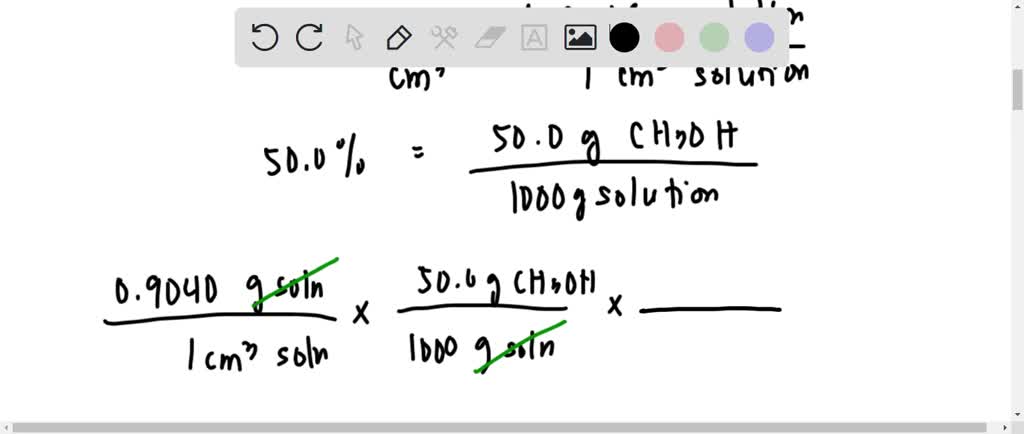
SOLVED: What is the molarity of a 50.0% by weight aqueous solution of methanol (CH3OH)? 8, The density of the solution is 0.9040 g/cm3. The molar mass of methanol is 32.04 g/mol

Molar Heat of Combustion, Definition, Equation & Formula - Video & Lesson Transcript

⏩SOLVED:The density of acetonitrile (CH3 CN) is 0.786 g / mL and the…

Twisted-Planar-Twisted expanded porphyrinoid dimer as a rudimentary reaction-based methanol indicator
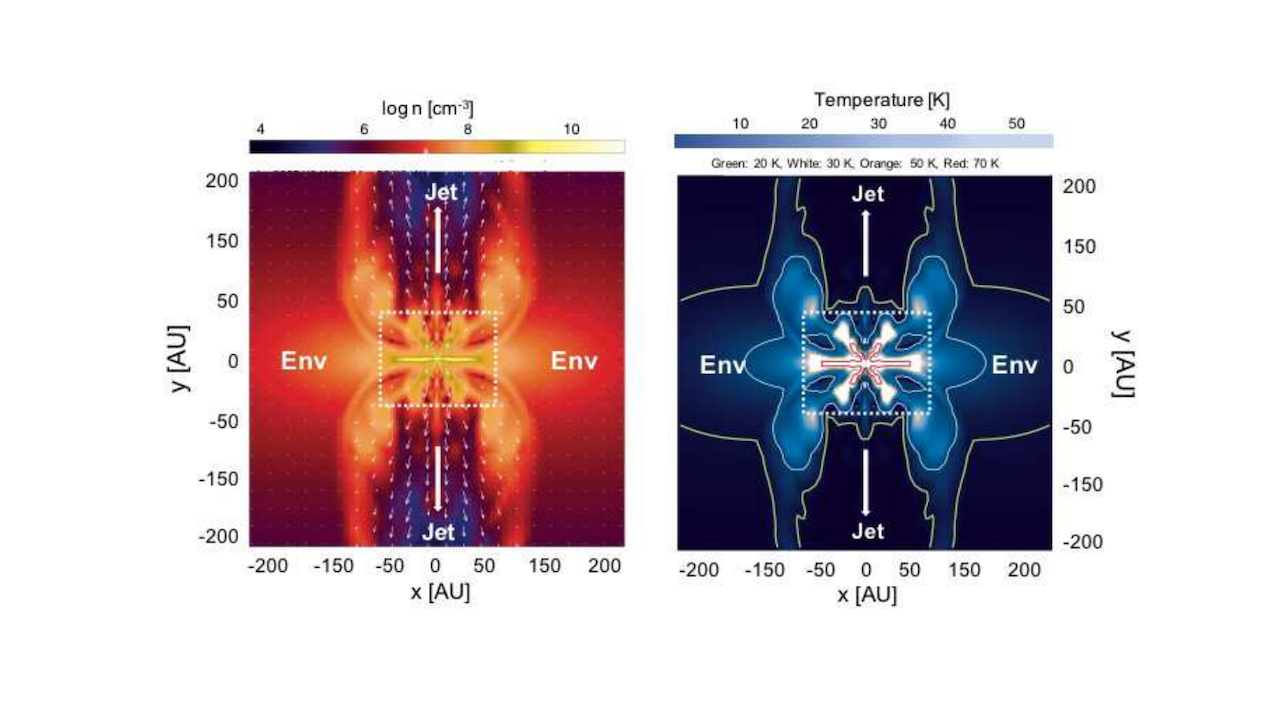
First Observations Of Warm And Cold Methanol In Class 0/I Proto-brown Dwarfs - Astrobiology

Calculate the molarity (M) and molality (m) of 16% aqueous methanol (CH_(3) OH) solution by volu
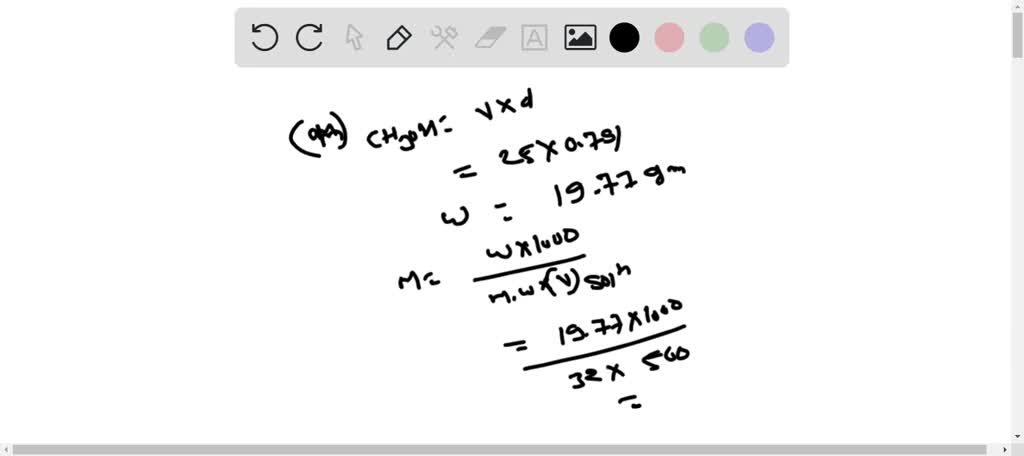
SOLVED: 1-A. A solution with a final volume of 500.0 mL was prepared by dissolving 25.00 mL of methanol (CH;OH, density 0.791 4 glmL) in chloroform. (a) Calculate the molarity of methanol

Methanol, CH3OH

SOLVED: Bridgman obtained the following volume ratios V/Va for methanol, where Vo = 39.6 cm³ is the molar volume at 0°C and pressure of kglem?. (kg cm³) V(20°C) V(50°C) 500 1000 0.9530

Methanol, Structure, Density & Molar Mass - Video & Lesson Transcript

Quick Video: Density calculation of methanol